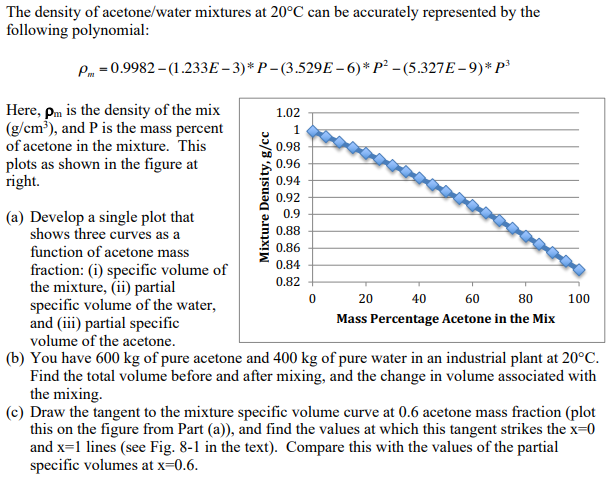
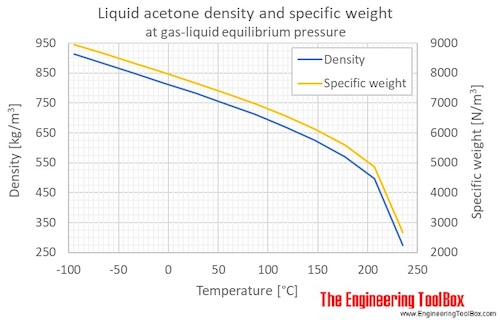
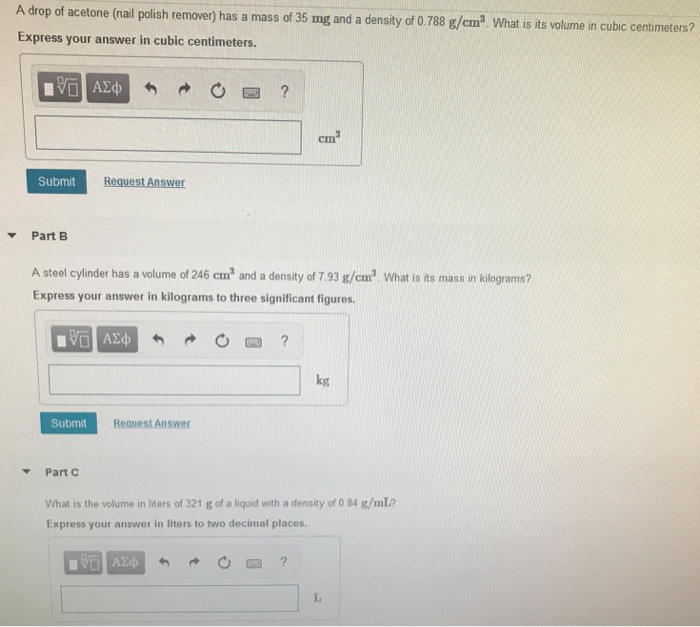
SOLVED: The density of acetone is 791 kg/m³. This is equivalent to 3.60 x 10³ lb/L 1.74 x 10³ lb/L b) 3.60 x 10² lb/L 1.74 x 10⠴ lb/L A solid

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram

OneClass: Acetone is a common solvent and has a density of 0.7899 g/mL. What volume of acetone, in mL...

SOLVED: An aqueous solution of acetone (CH3COCH3) with a molality equal to 1.29 mol/kg has a density equal to 0.990 g/mL. What is the volume molar concentration?