Diketene a Privileged Synthon in the Synthesis of Heterocycles. Part 2: Six-Membered Ring Heterocycles - ScienceDirect

Arylation Using Sulfonamides: Phenylacetamide Synthesis through Tandem Acylation–Smiles Rearrangement | Organic Letters
![Early Amidation Approach to 3-[(4-Amido)pyrrol-2-yl]-2-indolinones | The Journal of Organic Chemistry Early Amidation Approach to 3-[(4-Amido)pyrrol-2-yl]-2-indolinones | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/jo034304q/asset/images/large/jo034304qn00001.jpeg)
Early Amidation Approach to 3-[(4-Amido)pyrrol-2-yl]-2-indolinones | The Journal of Organic Chemistry
![EP2264027A1 - Process for the preparation of N-[2-(Diethylamino)ethyl]-5-[(5-fluoro-1,2-dihydro-2-oxo-3H-indol-3-ylidene) methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide - Google Patents EP2264027A1 - Process for the preparation of N-[2-(Diethylamino)ethyl]-5-[(5-fluoro-1,2-dihydro-2-oxo-3H-indol-3-ylidene) methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide - Google Patents](https://patentimages.storage.googleapis.com/66/70/14/db228d1e9562e6/imgb0018.png)
EP2264027A1 - Process for the preparation of N-[2-(Diethylamino)ethyl]-5-[(5-fluoro-1,2-dihydro-2-oxo-3H-indol-3-ylidene) methyl]-2,4-dimethyl-1H-pyrrole-3-carboxamide - Google Patents

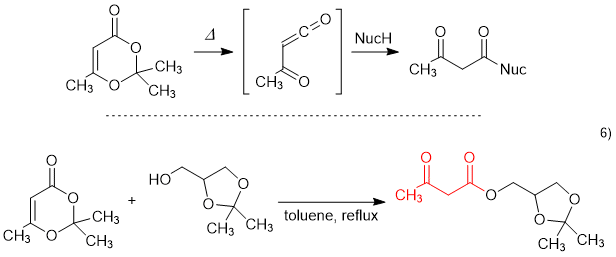
2,2,6-Trimethyl-1,3-dioxin-4-one, cont. up to ca 6% acetone, Thermo Scientific Chemicals | Fisher Scientific

Effect of ketene additive and Si/Al ratio on the reaction of methanol over HZSM‐5 catalysts - Hassanpour - 2018 - Applied Organometallic Chemistry - Wiley Online Library

Bond formations by intermolecular and intramolecular trappings of acylketenes and their applications in natural product synthesis. - Abstract - Europe PMC

Rapid, quantitative, solvent-free synthesis of medium-ring diaza heterocycles from diketene–acetone adduct and diamines - ScienceDirect

End group modification of polyamide-6 in supercritical and subcritical fluids: Part 3: Amine end group modification with diketene and diketene acetone adduct in CO2 - ScienceDirect

Molecules | Free Full-Text | Elagolix Sodium Salt and Its Synthetic Intermediates: A Spectroscopic, Crystallographic, and Conformational Study
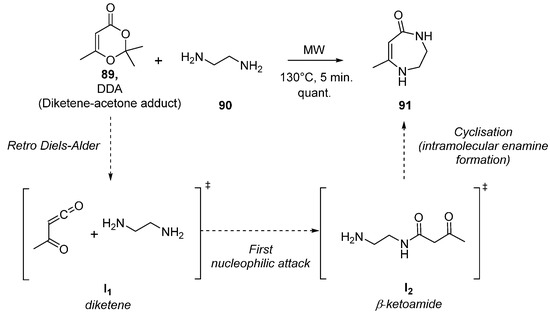
Molecules | Free Full-Text | Microwave-Assisted Syntheses of Bioactive Seven-Membered, Macro-Sized Heterocycles and Their Fused Derivatives

![2,2,6-trimethyl-m-dioxin-4-one - Optional[1H NMR] - Spectrum - SpectraBase 2,2,6-trimethyl-m-dioxin-4-one - Optional[1H NMR] - Spectrum - SpectraBase](https://spectrabase.com/api/spectrum/9pPIMRR7F5H/partial.png?h=214.875&ph=true&w=382)